by Brian Shilhavy
Editor, Health Impact News
As we reported over the weekend, the new plandemic being planned and rolled out to instill fear and sell more vaccines is the “monkeypox” virus.
Belgium is the first country to introduce a mandatory 21-day monkeypox quarantine for those who contract the virus, after three cases were recorded in the country. (Source.)
Vaccines that are already approved by the FDA already exist for smallpox, and those same vaccines, the public is being told, will protect people from monkeypox as well.
The CDC announced today that they are releasing smallpox vaccines for new cases of monkeypox in the U.S., which to date total ONE case and few others “suspected.”
Officials for the Centers for Disease Control and Prevention (CDC) on Monday said the agency is releasing doses of a smallpox vaccine in response to the few recent cases of monkeypox that have been detected in the U.S. (Source.)
Since these smallpox vaccines already exist, I did a search in the government VAERS (Vaccine Adverse Event Reporting System) database to see how many injuries and deaths have been reported following smallpox vaccinations, since smallpox has allegedly been eradicated in the U.S. for over 40 years now, with no known cases resulting in deaths or injuries during the past 40+ years.
Smallpox vaccines have caused 5,755 cases to be filed including 18 deaths, 91 permanent disabilities, 3,088 visits to the emergency room, and 851 hospitalizations, for a disease that no longer exists. (Source.)
And this is not a vaccine that has been given to many people in the past. It is not a vaccine that is in the CDC vaccine schedule for children, and in the statistics provided by the Health Resources & Services Administration website for total vaccine doses administered between 2006 and 2014, it is not listed there either.
According to the CDC, the smallpox vaccine has only been recommended “for specific populations at high risk of occupational exposure to orthopoxviruses.” (Source.)
So apparently these reported cases of injuries and deaths following smallpox vaccines are from a very small amount of smallpox vaccines given out during that past 40+ years.
There are two smallpox vaccines approved by the FDA (ACAM2000® and JYNNEOSTM), and a third one (Aventis Pasteur Smallpox Vaccine) that “is an investigational vaccine that may be used in a smallpox emergency under the appropriate regulatory mechanism (i.e., Investigational New Drug application [IND] or Emergency Use Authorization [EUA]).” (Source.)
One of these FDA approved smallpox vaccines, Jynneos, was just approved in September of 2019, and allegedly covers monkeypox in addition to smallpox. It is produced by Bavarian Nordic, and in 2019 the U.S. Government gave them $1.8 billion to secure 28 million doses to the “national stockpile” to “enhance domestic biodefense and global health security.”
These doses were apparently added to this “national stockpile” before the FDA even approved them. (Source.)
It is from this “stockpile” that the CDC is now releasing some to the public to deal with the “outbreak” of monkeypox in the U.S. that has infected one person.
Jynneos and Bavarian Nordic are not even listed yet in VAERS, which means this is an UNTESTED vaccine that is about to be distributed to people in the U.S. based on another false “pandemic.” All it currently has is FDA approval based on pre-launch studies, but apparently no real world experience of using it in the public.
Smallpox Vaccines Carry a LIVE Virus
Since we now have the evidence of massive fraud and criminal intent to commit harm and murder with the roll-out of the COVID-19 vaccines, it is wise that we take a very skeptical view of this new action by our nation’s corrupt government health agencies in rolling out these smallpox vaccines, which carry a live virus.
Since there are so few cases of smallpox at present, could the vaccines be the actual vehicle for spreading a new version of “monkeypox” to the rest of the population?
The CDC admits that people recently vaccinated with the existing FDA-approved smallpox vaccine can shed the virus in the vaccine to others.
Given the risk for vaccinia virus transmission from recently vaccinated persons through inadvertent inoculation, nonemergency use of ACAM2000 is also contraindicated in persons with household contacts with a history or presence of atopic dermatitis, other active exfoliative skin conditions (e.g., eczema, burns, impetigo, varicella zoster, herpes, severe acne, severe diaper dermatitis with extensive areas of denuded skin, psoriasis, or Darier disease [keratosis follicularis]); conditions associated with immunosuppression (e.g., HIV/AIDS, leukemia, lymphoma, generalized malignancy, solid organ transplantation, or therapy with alkylating agents, antimetabolites, radiation, TNF inhibitors, or high-dose corticosteroids [i.e., ≥2 mg/kg body weight or 20 mg/day of prednisone or its equivalent for ≥2 weeks], hematopoietic stem cell transplant recipients <24 months post-transplant or ≥24 months, but who have graft-versus-host disease or disease relapse, or autoimmune disease [e.g. systemic lupus erythematosus] with immunodeficiency as a clinical component); household contacts aged <1 year; and household contacts who are pregnant (recommendation category: A, evidence type 2 [Box]). Household contacts include persons with prolonged intimate contact with the potential vaccinee (e.g. sexual contacts) and others who might have direct contact with the vaccination site or with potentially contaminated materials (e.g., dressings or clothing). ACIP also does not recommend nonemergency vaccination with ACAM2000 for children and adolescents aged <18 years.
You can read about all the other Precautions and Contraindications for this FDA-approved smallpox vaccine here on the CDC website.
And guess what one of those side effects is that shares a common side effect with the COVID-19 vaccines?
Myopericarditis has also been associated with ACAM2000 and is estimated to occur at a rate of 5.7 per 1,000 primary vaccinees based on clinical trial data. (Source.)
Yes, weakened hearts resulting in heart disease is also a side effect of the smallpox vaccines.
Here is the age breakdown from the reported injuries and deaths in VAERS following smallpox vaccines: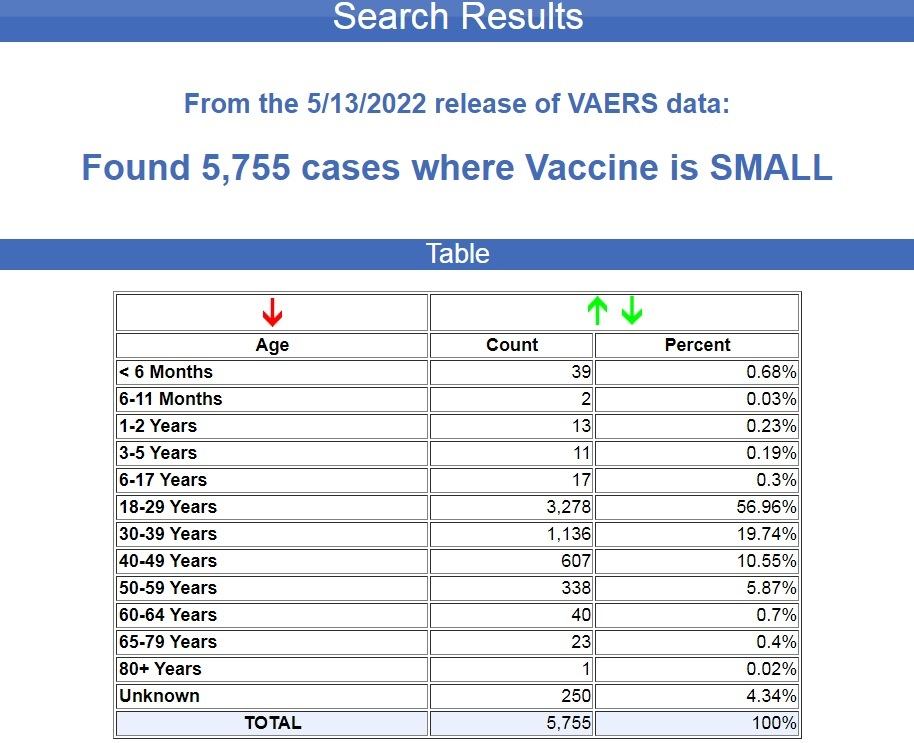
As you can see, about 77% of the cases were reported among young people between the ages of 18 and 39