By Jessica Rose
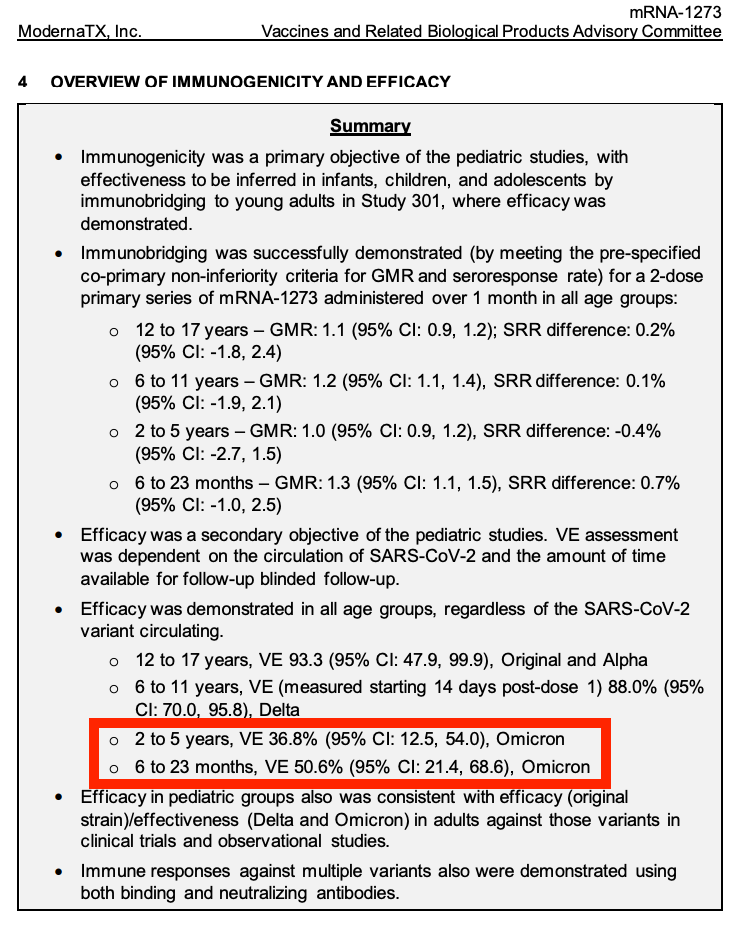
As most of you probably know, in a few days, the VRBPAC meeting will happen again. This time around, they have decided, ahem, will decide, on whether infants (6 months-4 years) should be injected with the COVID-19 injectable products. Yes, the same products with proven lamentable efficacy by their own admission (36.8% in 2-5 year olds), and horrific safety profiles and you will see.
Please go to this document entitled: “Vaccines and Related Biological Products Advisory Committee June 14-15, 2022 Meeting Presentation”. Released just in time is the document entitled: “mRNA-1273 PRIMARY SERIES 6 MONTHS TO 17 YEARS SPONSOR BRIEFING DOCUMENT VACCINES AND RELATED BIOLOGICAL PRODUCTS ADVISORY COMMITTEE MEETING DATES: JUNE 14–15, 2022”.
Alright I want to be succinct here. Please head to Page 43 of the Moderna document (the second link) for the OVERVIEW OF IMMUNOGENICITY AND EFFICACY. This will be all I have to say about efficacy and then I will pick on safety. It’s not that I think the Vaccine Efficacy measurement holds much weight anyway, but let’s assume that their measurement is adequate, for argument’s sake/ Even though we’re not allowed to argue. How in the world can they publish a 36.8% efficacy rate in 2-5 year olds and claim to have any justification whatsoever to inject children in this age group? The FDA Limit of Efficacy maintains that efficacy must be 50% over placebo to be approved for use.

Now let’s head to Table 26 on page 63. This was the first Table that caught my eye. It caught my eye because 20.5% of all 12-17 year olds in the drug arm (mRNA-1273) experienced and reported an adverse event (AE) within 28 days of injection. Of these, they even admit to 12.6% being ‘related to vaccination’. This is compared with 15.9% of 12-17 year olds on ‘placebo’ experiencing and reporting an AE.

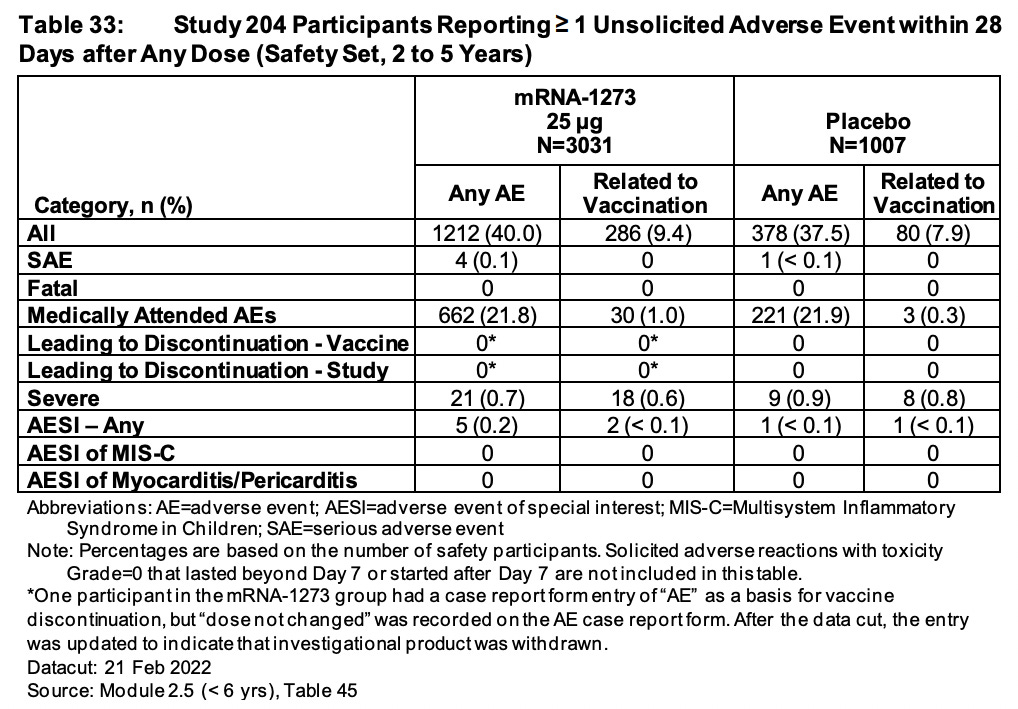
Now let’s head to Table 29 on page 72. Here we see 29.6% of all 6-11 year olds in the drug arm (mRNA-1273) experienced and reported an adverse event (AE) within 28 days of injection. Of these, they even admit to 10.6% being ‘related to vaccination’. This is compared with 25.1% of 6-11 year olds on ‘placebo’ experiencing and reporting an AE. I find this very strange. I still have a very strong suspicion that these ‘placebos’ are not saline and rather empty LNPs. Even if we take their 5% rate of AEs related to vaccination, how do they explain that?
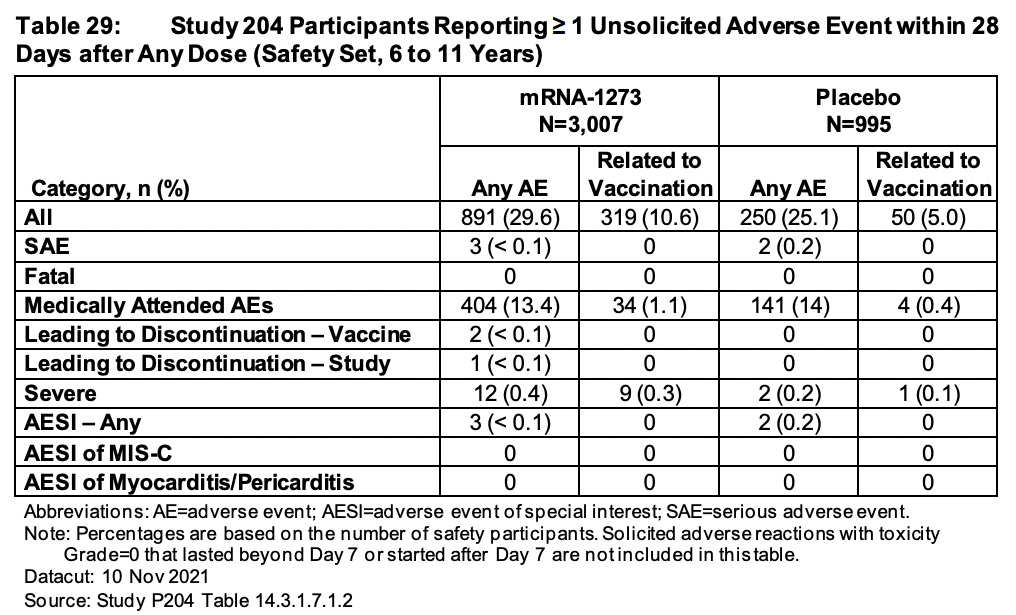
Now let’s head to Table 33 on page 82. Here we see 40% of all 2-5 year olds in the drug arm (mRNA-1273) experienced and reported an adverse event (AE) within 28 days of injection. Of these, they even admit to 9.4% being ‘related to vaccination’. This is compared with 37.5% of 2-5 year olds on ‘placebo’ experiencing and reporting an AE. Again, I find this high rate very strange. By the way, have you guys noticed that the cohort balance is ~2:1, ~3:1, ~3:1 in age groups 12-17, 6-11 and 2-5, respectively, with regard to mRNA-1273 and placebo. Again, why? Is this about your ‘ethics’ and how you justify injecting the unblinded clinical trial participants because ‘they deserve the vaccine’?
Now let’s go back to Table 27. 55.8% of the 12-17 year old children experienced and unsolicited AE of which 15% was claimed to be caused by the injection. So for an absolutely obsolete product (93.3% ‘VE’ in the context of Original and Alpha?), parents are expected to inject their children with something with an undisclosed ingredient list that, in 55.8% of children, is associated with an AE?

Now let’s go back to Table 38. 49.3% of the 6-23 months year old children experienced and unsolicited AE within 28 days of injection, of which 16.6% was claimed to be caused by the injection. So let’s look at that 0.5% of all AEs being serious thing. That doesn’t sound like much, right? I mean Walensky and Fauci would look at that and say, ‘woohoo!’. 0.5% = 5/1000. Maybe they should start expressing the rate in this way so that people remember that we’re talking about helpless infants. So if the public was informed that Moderna found that 1/200 children ages 6 to 23 months experienced a severe adverse event within 28 days of injection, I mean, like, at the injection site door, how many people would still go through with injecting their baby?
And by the way, what exactly is the difference between Severe and Serious? Because 1.2% of all 6-23 month olds experienced a severe AE. That’s a 1/83 chance of experiencing a severe AE.
I chose this table because it refers to babies but the rate is even worse for 12-17 year olds with 1/125 suffering a serious adverse event. No time frame reported. Maybe some of you lovelies could make some posters and pin them to injection site doors.

So these are just a select few Tables that stuck out for me. By the way, do any of you find it appalling that the highest number of trial participants was 2,486, 3,007, 3,031 and 1,761, for age groups 12-17, 6-11, 2-5 and 6 to 23 months, respectively.
There is still time to file a comment but you guys should know that of the 126,947 public comments made in just 6 days, they are hiding them from the public. Make your comments anyway. You can do that here until June 14th, 2022 to do so. Use Docket No. FDA-2022-N-0904.
Here. I made this. Please print and post everywhere. Schools. Parks. Clinics. Give to MDs. Make them read the bloody document. INFORM THEM AND HOLD THEM ACCOUNTABLE in case of injury.