We have access to 150 documents to date. Here’s a first-cut analysis of what we found.
by Steve Kirsch
CAUTION: This document is still being edited and fact checked and is likely to contain errors. Use at your own risk. It has NOT been pushed to my subscribers. If you find a mistake, let me know in the comments. For example, Point #5 is not right since immunity isn’t correlated to this and patients were given different doses.
I am writing this article with Kyle Beattie. Kyle was the author of the 145 country study that nobody was able to refute that showed that the more you vaccinate, the higher the COVID infection and death rate that I wrote about on January 8, 2022.
About the documents
We’ve been hard at work going through the Pfizer documents that were released on March 1, 2022.
Pfizer has done a series of drops starting with documents on the 18th of November, 2021 and all the way up to first of March this year.
There are 150 documents so far comprising an estimated 50,000 pages. The documents are all posted here on this third-party portal (it is not run by Pfizer):
So far, they have given us some of their internal code dictionaries, which correspond with various other documents, studies, and spreadsheets and will take some time in order to analyze completely (they use SAS mostly). They have also provided some documents they sent and received from the FDA as well as some of their results of their own clinical animal and human trials. This is a summary of the interesting items we have found so far.
Limitations of our analysis
Since I have a limited staff on this project (me and Kyle), we won’t be able to do everything that others will cover. I am sure that the mainstream media and also the medical community will be all over these documents exposing the corruption in great detail.
OK, I was just kidding. The truth is that the only people who are going to let you know what is really going on in these documents are the people that the mainstream media calls “misinformation spreaders.” Everyone else is going to pretend like nothing is wrong.
Key conclusion: their data doesn’t support their claims
Pfizer’s own data does not support the claim their COVID-19 vaccines are “safe and effective.”
Pfizer seemingly lied to the FDA by claiming their vaccines prevent COVID-19, and the FDA and mainstream media then dutifully lied to the American people repeating this false claim.
Things the documents tell us now
Here are the some of the issues we will cover in this review. More to come as we learn more:
- Pfizer claimed repeatedly in their documents to the FDA that their vaccine would “prevent” COVID-19.
- Pfizer knew the injection’s adverse effects would increase with more injections of continuing boosters.
- Pfizer knew their injections did not stay at the injection site.
- Pfizer knew that the vaccinated group reported far more systemic adverse events than the placebo group.
- Pfizer knew that the efficacy of the vaccine waned very quickly over time; by as much as 50% in as little as 1 month after the second dose. How come we weren’t warned about that???
- Pfizer defended VAERS (because they didn’t want extra reporting cost burdens).
- There are six individuals that signed up for two different clinical trials at two different sites which is really odd.
- Pfizer knew vaccinated individuals could still catch COVID-19 and test positive.
- There are 1,448 pages comprising 9,704 individual subjects who were excluded from the trials. There isn’t enough detail to know why.
- Pfizer paid $2,875,842.00 for their application to the FDA. This is more of a point of information for now.
- It is troubling that Pfizer redacts information that is not proprietary that would be very helpful in assessing the data such as the number of doses administered in the ADVERSE EVENTS OF SPECIAL INTEREST” (AESI) document (aka the 5.3.6 document).
1. Pfizer claimed repeatedly in their documents to the FDA that their vaccine would “prevent” COVID-19
In this document, they claimed on Page 1, Box 15A: that their application was for a product with the following Proposed Indication for Use: “Active immunization to prevent COVID-19 caused by SARS-CoV-2 in individuals ≥ 16 years of age”.
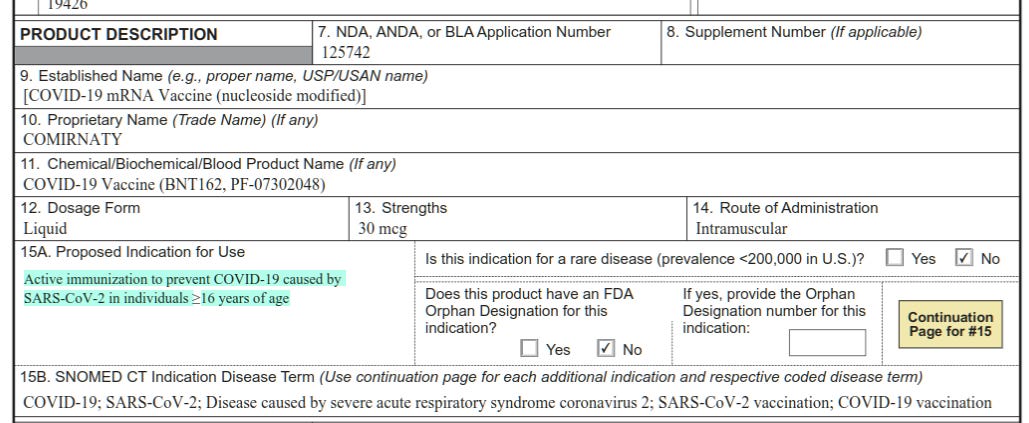
Here on Page 3, they make the same claim again, this time in a waiver document that is described in more detail in point number 6. They state, “The proposed indication that is the subject of this initial BLA application is active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older” [emphasis added].
Also in this document on page 1 they again claim that, “The proposed indication is active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older. [emphasis added]”

Did Pfizer defraud the FDA by claiming the vaccine they were proposing would “prevent” COVID-19?
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M1_356h.pdf
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M1_waiver-req-designated-suffix.pdf
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_22_introduction.pdf
2. Pfizer knew the injection’s adverse effects would increase with more injections of continuing boosters
Specifically, on page 24 of this document, they state that, “Local reactions were observed in male and female animals dosed IM with BNT162b2 (V8). The incidence and severity of the reactions were higher after the second or third injections compared with the first injection. The majority of animals had very slight edema or rarely slight erythema after the first dose. After the second or third dose, the severity of edema and erythema increased up to moderate or rarely, severe grades. [emphasis added]”
Clearly they knew the side effects were dose dependent and thus they knew the vaccines were causing these effects.
They also knew that these adverse effects could include such things as muscle necrosis and increased spleen size and weight.
They state, “BNT162b2 (V8)-related higher absolute and relative (to body) spleen weights (up to 1.62x controls) were evident and correlated with the macroscopic observation of increased spleen size…Injection site inflammation was associated with moderate edema, mild myofiber degeneration, occasional muscle necrosis, and mild fibrosis” (page 25).
These terrible reactions, particularly the necrosis, we have seen evidence of in the medical literature after administration of these vaccines in the wider public. Indeed, their own animal studies indicated as much.
Funny, when I was vaccinated they never warned me about any of this. I don’t recall Fauci or Walensky mentioning it either. Do you?
See the following studies for discussions of various forms of necrosis after COVID-19 vaccine administration:
Lal, N., Khan, S. A., Pillai, A. R., & Khan, S. A. (2021). COVID vaccine related lower limb gangrene: the first case report. International Surgery Journal, 8(10), 3204-3207. https://dx.doi.org/10.18203/2349-2902.isj20214026
Kuzumi, A., Yoshizaki, A., Chiba, K., Mitsuo, S., Matsuda, K. M., Norimatsu, Y., … & Sato, S. (2021). Genital necrosis with cutaneous thrombosis after COVID‐19 mRNA vaccination. Journal of the European Academy of Dermatology and Venereology. https://doi.org/10.1111/jdv.17837
Mishra, S. B., Mahendradas, P., Kawali, A., Sanjay, S., & Shetty, R. (2021). Reactivation of varicella zoster infection presenting as acute retinal necrosis post COVID 19 vaccination in an Asian Indian male. European journal of ophthalmology, 11206721211046485.
Iwai, S., Takayama, K., Sora, D., & Takeuchi, M. (2021). A Case of Acute Retinal Necrosis Associated with Reactivation of Varicella Zoster Virus after COVID-19 Vaccination. Ocular immunology and inflammation, 1-3. https://doi.org/10.1080/09273948.2021.2001541
Gruenstein, D., & Levitt, J. (2021). Skin necrosis at both COVID-19 vaccine injection sites. JAAD Case Reports, 15, 67-68. https://www.jaadcasereports.org/article/S2352-5126(21)00510-5/fulltext
Ramessur, R., Saffar, N., Czako, B., Agarwal, A., & Batta, K. (2021). Cutaneous thrombosis associated with skin necrosis following Oxford‐AstraZeneca COVID‐19 vaccination. Clinical and Experimental Dermatology. https://dx.doi.org/10.1111%2Fced.14819
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_24_nonclinical-overview.pdf
3. Pfizer knew their injections did not stay at the injection site
As I pointed out back in May of 2021, the injection doesn’t stay at the injection site.
The exact same data is included in the released documents as Byram Bridle obtained in May via FOIA from the Japanese government.
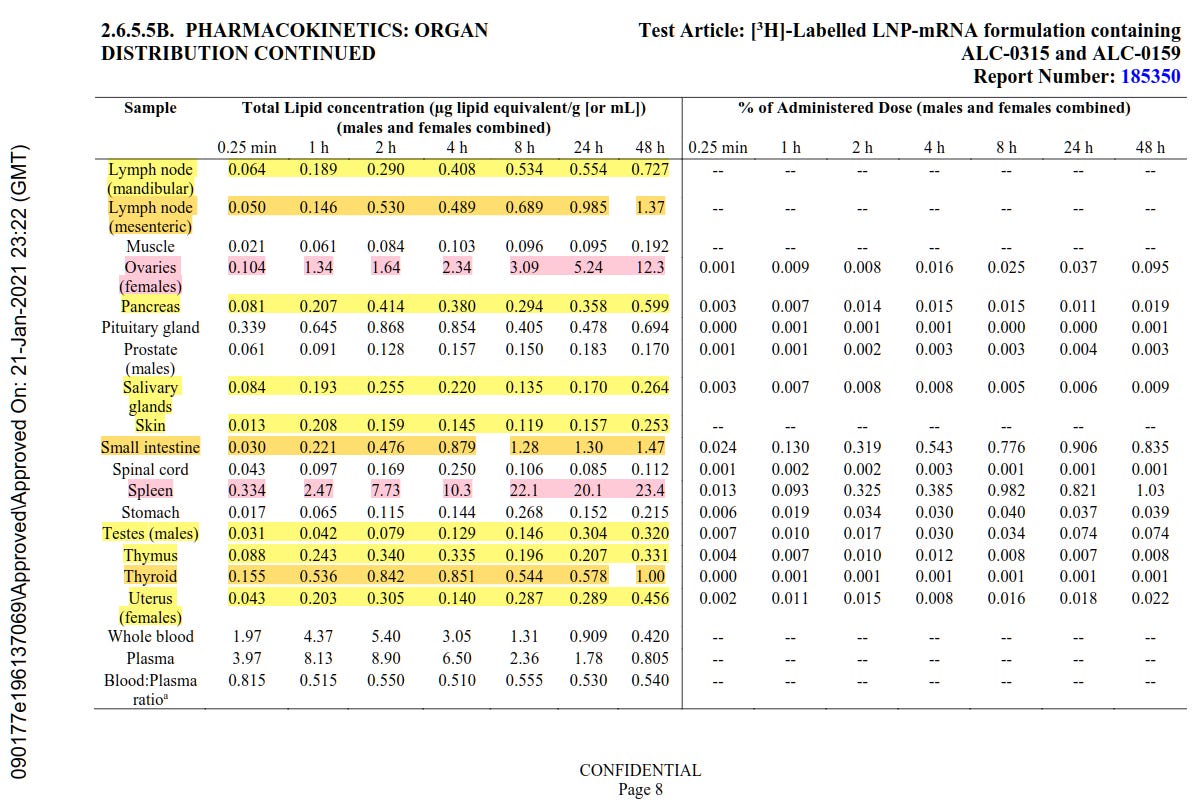
Indeed their own data from animal trials shows that with just one dose over a 48 hour period the vaccine quantity decreases from the injection site and increases substantially in the ovaries, liver, and spleen in particular, but also in adrenal glands, bladder, bone, bone marrow, eyes, large intestine, lymph nodes, pancreas, salivary glands, skin, small intestine, testes, thymus, thyroid, and the uterus.
To be specific, 0.09% of the injection ends up in the ovaries, 1.03% of the injection ends up in the spleen, and around 16.2% of the injection ends up in the liver after 48 hours (page 7 and 8).
Please see the tables on page 8 of this report, which refer to Report Number: 185350, which is referenced on page 2.
page 2
Yellow highlighting means the amount of μg lipid equivalent/g of the Pfizer injection increases rather than decreases over the 48hr period they observed. Orange means same as yellow but moderately high compared with others. Red means same as yellow but it is very high compared with all others. Green is where the injection started, and where they claimed it would stay. It clearly decreases over time and shows up other places in your body particularly the liver, ovaries, and spleen.
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_26_pharmkin-tabulated-summary.pdf
4. Pfizer knew that the vaccinated group reported far more systemic adverse events than the placebo group
Pfizer’s own reactogenicity data shows that the people receiving the injections of BNT162b2 (30 μg) are anywhere from twice to 25 or more times as likely to have severe systemic events compared with the placebo group.
A quick glance through this data shows just how badly the vaccine affected people compared with the control group. For example, within seven days after each dose, twice as many people (23%) in the vaccinated group suffered systemic events compared with the placebo group (11.3%), while severe fever was noted in the vaccinated group 14 times as much as the placebo group.
page 35
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M5_c4591001-T-S-final-reacto-tables-track.pdf
5. Pfizer knew that the efficacy of the vaccine waned very quickly over time
While the mainstream media did the best they could at selling the Pfizer vaccine as “safe and effective,” Pfizer’s own data as early as September 19, 2020, showed neither of those things.
In particular, the vaccine efficacy, even for the short period for which it was studied, showed that it waned by as much as 50% in as little as 1 month after the second dose (See S1-Binding and RBD-Binding IgG Levels in the following table).
This information was hidden from the public and was widely denounced as medical misinformation until the “rare breakthrough cases” became so common that they could no longer be denied.
Turns out, they knew the whole time.
They just didn’t tell us for some reason and neither did the FDA or CDC. It was probably just an oversight.
30 μg dose rate
6. Pfizer defended VAERS
Pfizer applied for an FDA waiver to not have to record certain safety data on the injections because they claimed VAERS was the system that would tell us if there were safety issues with the injections. Then the mainstream media disparaged VAERS data for the past year and acted as if it was not as significant as it really was.
In reality, on page 3 and 4 of their waiver request, Pfizer says VAERS is a “robust” system that is “designed to detect safety concerns with vaccines.” They even used this excuse for their suffix waiver claiming they didn’t need to have an FDA designated suffix that would help “ensure safe dispensing practices and optimal pharmacovigilance” because the VAERS system was “adequate” and that any other safety reporting requirements would be “redundant and burdensome.”

page 4
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M1_waiver-req-designated-suffix.pdf
When they don’t want to do extra work, it is amazing how reliable and accurate VAERS is. But when there are adverse safety signals, then VAERS is junk.
Get it?
7. There are six individuals that signed up for two different clinical trials at two different sites
We have some real go-getters in this country, we had half a dozen people sign up to be guinea pigs for not just one experimental injection study, but two! According to page 20 of this Pfizer document, “6 Subjects were enrolled into the study more than once”. Unfortunately, their atta-boy attitude did not help the study and Pfizer was forced to eliminate their results stating that, “These subjects will not be included in any analyses.”
That’s comforting, but how they heck did they get past the enrollment process?
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M5_c4591001-A-adrg.pdf
page 20
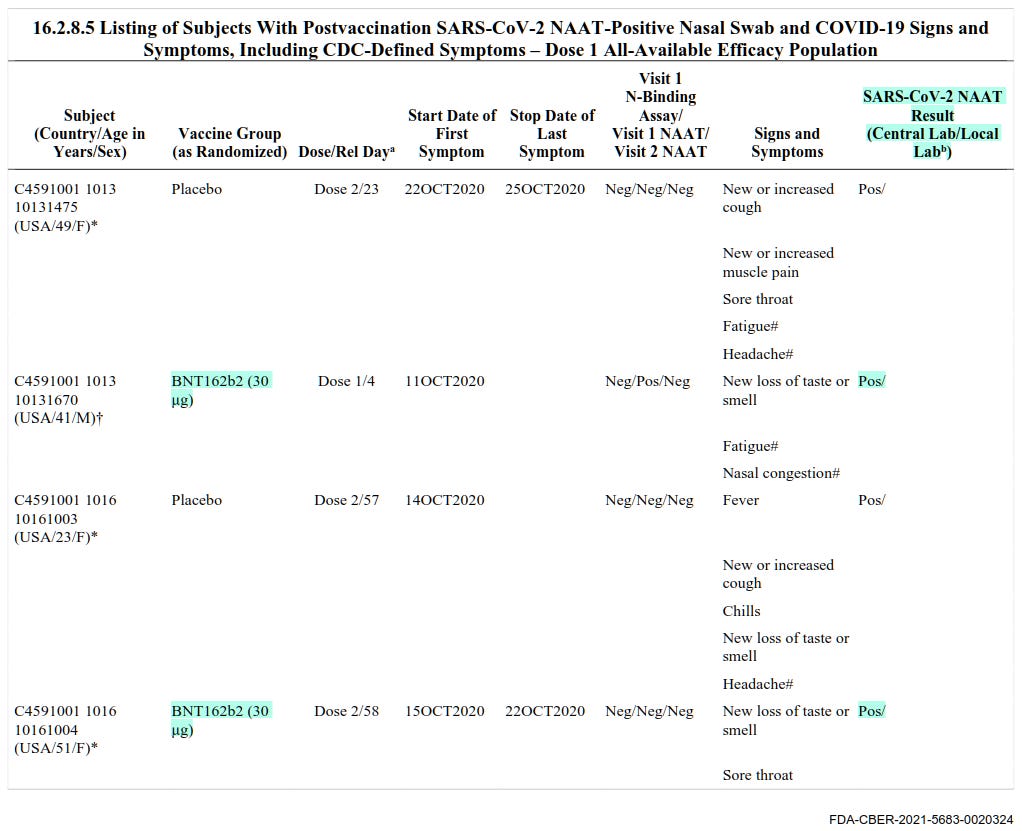
8. Pfizer knew vaccinated individuals could still catch COVID-19 and test positive
In this document, Pfizer provides tables of all the different conditions under which people got sick with COVID-19 after the vaccination or placebo was administered. Overall, 12% of reported positive and/or symptomatic cases are individuals who have been vaccinated with either one or two doses of Pfizer (BNT162b2, 30 μg). This means that Pfizer’s claim in their other documents that this vaccine prevents SARS-CoV-2 infection is demonstrably false, and they knew it.
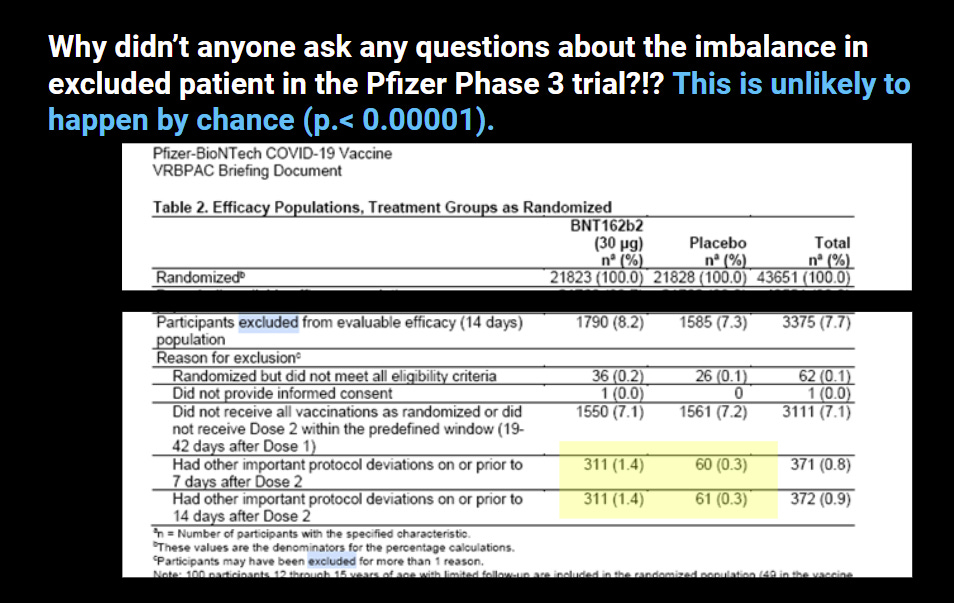
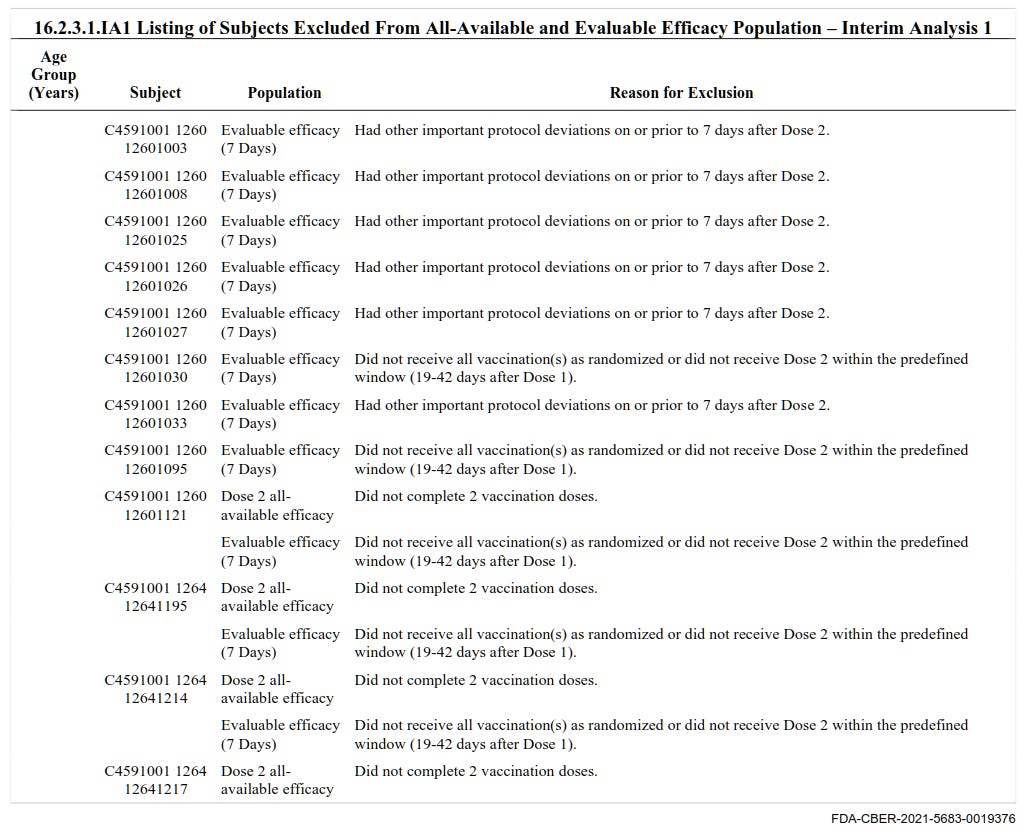
9. There are 1,448 pages comprising 9,704 individual subjects who were excluded from the trials
These people were excluded from the trials for various reasons including not receiving Dose 2 within the predefined window, not receiving all vaccinations as randomized, or having important protocol deviations.
Remember, all the people who never went back for their second shots during 2021?
How many of these individuals in the clinical study didn’t go back for their second shot because of some reaction they may have had?
What we know from my earlier work is that the number of drop outs in the Phase 3 trail were impossibly high in the treatment group. It couldn’t have happened by “bad luck.”
Here’s the slide from an earlier slide deck I did (see slide 33, but the rest of the presentation is awesome too):
Also, this brings to mind the Ventavia Research Group whistleblower whose story was published in the British Medical Journal regarding “poor laboratory management, patient safety concerns, and data integrity issues”.
10. Pfizer paid $2,875,842.00 for their application to the FDA
If you were wondering what it cost Pfizer and what the FDA charged to evaluate this horrid series of reports and clinical studies included in their application, it was around $2.9 million dollars.
Source: https://phmpt.org/wp-content/uploads/2022/03/125742_S1_M1_userfee.pdf
Other analyses of the Pfizer data
- Dr. Been looks at the “5.3.6 document” which is the post-marketing adverse event data in this 1 hour YouTube video. This is the document with the famous: “LIST OF ADVERSE EVENTS OF SPECIAL INTEREST” (AESI) which has 9 pages of adverse events packed very tightly. My favorite comment on this video is this one: How is it possible that Dr Been, singlehanded, can clearly inform the public, while the US government fails every opportunity? That’s a good question.
Summary
We will be publishing more of this information as we have time to go through it with exact links to every page, but we’re sure other independent investigators will look through these documents as well. Specifically, we need people who are skilled in reading these types of pharmaceutical reports and anyone who has good skills with SAS databases.

